Transforming lives.* Incisionless therapy for movement disorders.

+30,000
Global
treatments**
Register here to receive invites to our webinars on the latest updates on focused ultrasound.
Essential Tremor1
Unilateral and staged, bilateral MR-guided focused ultrasound.
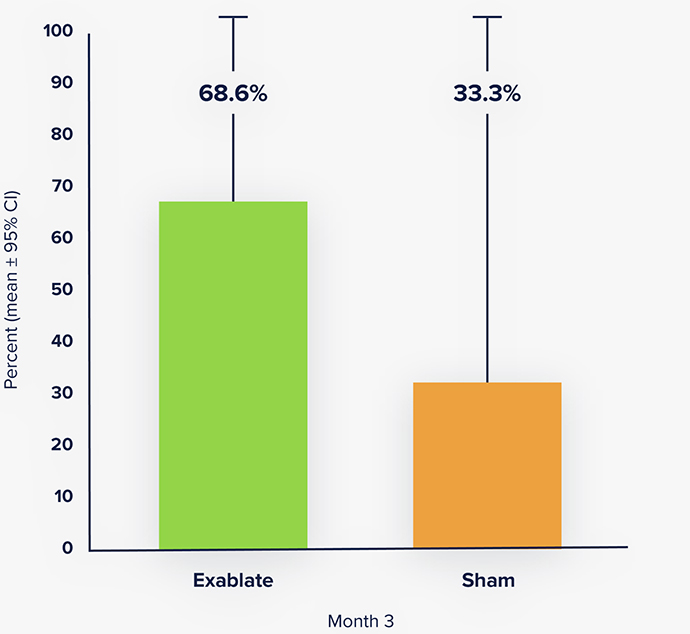
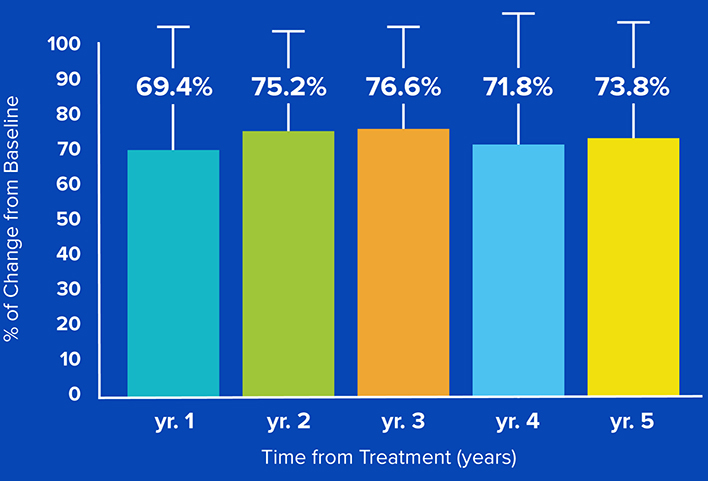
5-year study results demonstrate significant
and sustained tremorimprovement.
73.8% Improvement in tremor severity
at 5-years (CRST Part A).
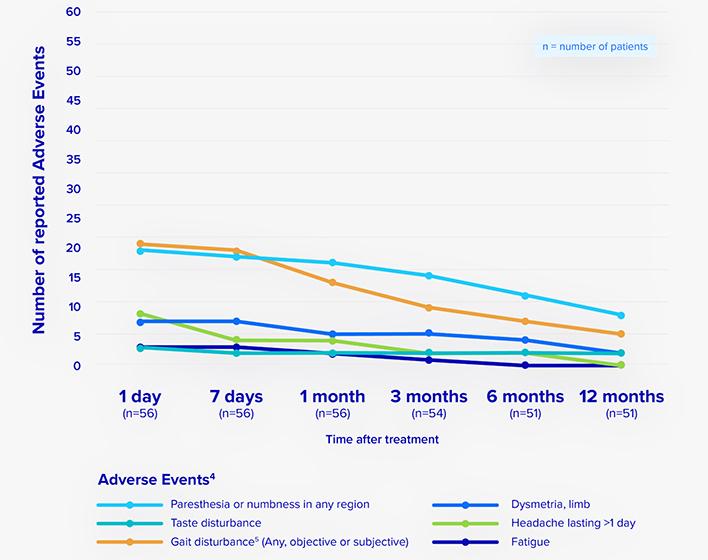
Overall, focused ultrasound has been shown to be safe for treating essential tremor with minimal risk.
Most adverse events were mild and resolved in 6 months.
For complete safety data: https://insightec.com/safety-information
Reported adverse events following unilateral focused ultrasound treatment for essential tremor.
Refer your patients for focused ultrasound.
Revolutionary treatment for essential tremor,
now approved to treat both sides.1
Parkinson’s Disease3
Unilateral MR-guided focused ultrasound for tremor dominant
Parkinson’s disease.
53%
improvement in
functional disability
(CRST part C).
40%
improvement in quality
of life (QUEST).
Tremor-motor function significantly improved (CRST A+B composite)
Parkinson’s Disease4
MR-guided focused ultrasound for motor complications of Parkinson’s disease.
Unilateral MR-guided focused ultrasound pallidotomy is indicated for patients over 30 with confirmed diagnosis of idiopathic Parkinson’s disease.
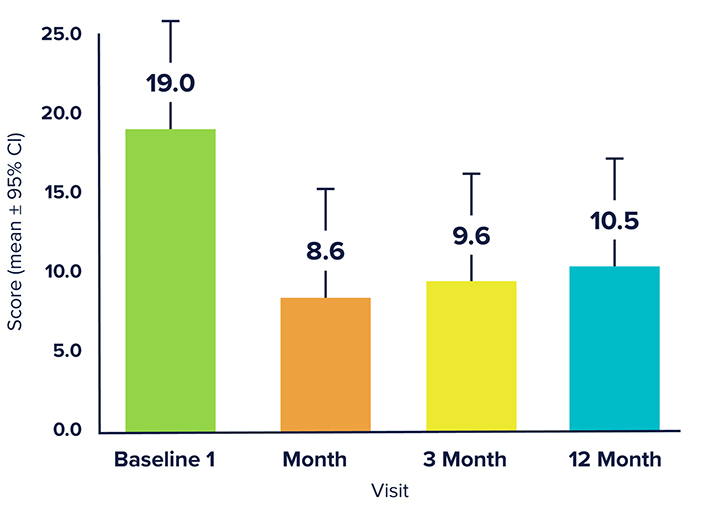
Significant improvement,
in motor complications and motor symptom severity following focused ultrasound treatment of the GPi.
26.4%
Improvement in MDS-UPDRS
Part III OFF at 3 months
46.1%
Improvement in
MDS-UPDRS Part IV
No severe or life threatening events.
Primary efficacy
responder analysis.